Content Reference:
https://www.youtube.com/watch?v=DjuK3NhEbLc I ignored the pain. That was a mistake.
I was diagnosed to be G6PD positive, essentially meaning that there was an enzyme not present in my blood. A very rare case, but then again with the permutations of the human gene, a little deformity is expected, given the individualistic nature of each human being. To explain it further, this enzyme, glucose-6-phosphate dehydrogenase, or G6PD, helps the red blood cells function normally. Its absence or deficiency can cause hemolytic anemia, usually after exposure to certain medications, foods, or even infections. Obviously, I was totally ignorant of either the fact that I had this condition or that it could prove to be fatal. It started with a dose of some pain reliever we normally pop in for everyday ailments like headache. The reactions it triggered in me were very mild at first. Pain in the lower abdomen area and shooting pain in the groin region. Coming from a relatively close knit family, I didn’t want to cause unnecessary stress to my parents and siblings, and thus foolishly tolerated the pain through sleepless nights. And then it got really bad. On the 4th night, I was urinating very dark red fluid, with a burning sensation. I put myself through that night. The whole day was lost, writhing in agony and trying to eat and drink, only to throw up. I was on liquids. Three nights of this red urine later, could not bear the pain anymore and asked my folks to take me to the hospital right away. It was in the middle of the night. The next early morning, we rushed to our family doctor, an hour’s drive away. The entire journey I can remember trying to remain conscious through unbearable pain. As soon as we reached the hospital, I could hear the doctor give some urgent orders around while I was moved hurriedly to an enclosed ward. A long pipe was inserted in through my mouth, leading the other end in a transparent plastic bottle. I was asked to pump my stomach by myself and try to push as much as liquid I could out through the pipe. All this with unbearable shooting pain and trying to remain conscious. I pushed my insides, with all the force and saw green gooey stuff rush through the pipe into the bottle. The pain lessened a wee bit. I was encouraged to push harder. To what seemed like forever I kept flushing my guts out, to fill nearly a litre of toxic formation. A few hours more, and maybe one of my kidneys would have to be taken off. It was a very close call. Seeing the green gooey toxic liquid, was making me want to puke more. Only my entire body was drained of fluid. The danger was not yet over. I was rushed to an A-grade hospital facility in Bombay, Hinduja Hospital. From the time, I was whisked in through to the emergency ward, I could feel some of myself feeling hopeful about my survival. I was immediately given a bed in the ICCU. My RBC count had fallen to all all-time low and very dangerously so. Four bottles of blood, and some 20 saline bottles in two days finally got me back to some normalcy. Funnily, it was my sense of humor and the inherent flirt in me which kept my spirits alive, and in turn my body. I had a sleep, wake, eat, clean schedule. And I had procured a pen and a paper pad to doodle. No visitors were allowed, so my family and friends only could see me through the glass window in the door. I was recuperating well, compared to the semi-conscious patients around me. In those 3 days, one of them passed away. It was a very real, wake up call. It is strange, how even with the inevitability of it, we are never prepared for it. Even it’s a someone whom you didn’t know. But the act of hearing the beeps on the monitor go off, and the nurse taking off all the plugs and pipes poking through the body, makes one realize the frailty of life and also the uncertainty of it. I was thankful to be alive and well. There were constant monitoring of my state and I didn’t mind being the ‘case study’. These were days when G6PD deficiency was not all that common and there weren’t too many live cases to study on. I was shifted to the general ward, where the monitoring continued. They tested my blood for every damn chemical and that’s when they came across this deficiency in mine. Seeing my averseness to allopathic medicines and popping pills, I was more comfortable with the needle pricks. Some people just have a different set of pain resisting profile. During this entire phase, there was a very senior doctor who was taking care of me. Dr.F.D.Dastoor. He was curious and patient and extraordinarily caring. He didn’t force me to take my pills, only the vitamins, and was okay with me skipping my sleeping pills in case I fell asleep reading. On my way to recovery, I had the fortune to talk to him on many an occasion. “70 percent disease, starts in the mind” these wise words stuck with me forever. From then on, and this is back in 1994, I have time and again, reinstated this magic mantra and it has worked. Twenty days later, I was discharged with a clean chit. I had successfully recuperated to my usual health, and as long as I stayed away from a list of medicines and pear (yes, the fruit) there seemed to be no reason for any kind off relapse on this anemic attack. In my final trips to the doctor, he gave me another simple wellness measure. “If, you are urinating clean and clear, your digestive system is in top condition and there is nothing wrong with your system”. On occasion, when I have due to lack of time, or just plain neglect, not had enough quantity of water, the yellowish coloration of the urine rings the warning bells loud in my head. And I hydrate myself, immediately. These two very simple and very basic rules, have kept me away from hospitals, except for a couple of fractures, (mild osteoporosis ). So, living isn’t all that difficult, if we can manage to keep the disease out of our head. To be healthy, stay happy and hydrated. BY SANTOSH DHEMRE Treatment: In most cases, individuals suffering from G6PD deficiency do not require treatment. Instead, drugs, chemical exposures and foods like fava or broad beans that are able to cause oxidant stress should be avoided. Hence, it is essential to identify and discontinue such substances in patients with G6PD deficiency. Treatment for anemia should be appropriately selected by the patient’s doctor and is influenced by factors such as overall health, medical history and tolerance for specific medication and procedures. However, red blood cell transfusions are rarely needed for G6PD deficiency anemia and may be used in severe cases of haemolytic anemia and neonatal jaundice (caused by access of bilirubin). Infants that suffer from prolonged neonatal (relating to newborns) jaundice due to G6PD deficiency may seek phototherapy with bili light (a light therapy tool) which induces the secretion of bilirubin by converting them into water soluble isomers through the use of light. In cases of non-spherocytic anemia or chronic hemolysis, patients should be given folic acid supplements which are taken on a daily basis. However, a geneticist and a haematologist should be consulted. Events of acute kidney injury may also require dialysis. However, as they say, Prevention is better than treatment. How can we prevent G6PD deficiency?Prevention:
Neonatal screening for G6PD deficiency is one of the best way to allow prevention and timely treatment. A study in southern Iran shows that the hospitalization rate of people with G6PD deficiency decreased after the neonatal screening program was practiced there. Symptoms such as neonatal hyperbilirubinemia and hemolytic anemia were able to be prevented and treated within a week of childbirth. Furthermore, people with a family history of G6PD deficiency conditions should seek genetic counselling for more knowledge and advice about the risk of passing on or inheriting the condition and go for genetic tests such as enzyme assays that measure enzyme activity of G6PD or sequence analysis of the coding region that cause G6PD deficiency to occur. G6PD Deficiency Is Associated With Significant Protection Against Severe, Life-threatening Malaria12/23/2014 A case-control study in two populations in Mali, West Africa has shown that glucose-6-phosphate dehydrogenase (G6PD) deficiency is associated with significant protection against severe, life-threatening malaria. Researchers from the US National Institute of Allergy and Infectious Diseases and the University of Bamako, Mali, led by Thomas E. Wellems, report the findings this week in the journal PLoS Medicine. G6PD deficiency is also known as "favism" after the Italian word for broad beans (fava) which cause a classic reaction when eaten by people with G6PD deficiency. In males, who have only one X chromosome, mutations in the gene for G6PD on the X chromosome cause G6PD deficiency. Females who have mutations on both X chromosomes will also be deficient. G6PD is an important enzyme in red blood cells (erythrocytes), the host cells for Plasmodium falciparum, the parasite that causes the most severe form of malaria. G6PD deficiency is associated with protection against malaria, notably in Africa where one form of G6PD deficiency (G6PD A-) is widespread. In the two populations of more than 3000 children studied in rural Mali where malaria is very frequent, G6PD deficiency in male and female children was associated with protection against severe, life-threatening malaria, but no effect was found in females who had just one abnormal gene. However, there was no significant difference in the numbers of parasites in the red blood cells of the various groups of children indicating that the deficiency does not work by stopping parasites from infecting the children. G6PD deficiency instead appears to mitigate disease processes set up by the parasitized cells in the bloodstream. The protection was confirmed by a combined analysis of these data and data from a previous study. Protection was most evident against cerebral malaria, the most frequent form of life-threatening malaria in these studies. These results reignite the debate about the relationship between G6PD deficiency and protection against malaria. They are particularly relevant in populations such as the one studied where, as the authors note, because "virtually all young children experience episodes of malaria, such protective hemoglobinopathies and erythrocyte polymorphisms offer a tremendous survival benefit when they prevent progression of uncomplicated malaria." Content reference:
http://www.sciencedaily.com/releases/2007/03/070313114502.htm Vasilli Kalisperas from Malvern in Worcestershire had a condition called kernicterus and it could have been treated with a light therapy or a blood transfusion but because of the carelessness of a trainee midwife who took care of him, she did not recognise the early symptoms of jaundice caused by the condition. Kernicterus is a condition whereby bilirubin, a yellowish substance found in bile, enters the central nervous system and causes neurological damage to the brain. Apparently, the effects of kernicterus was sped up by the condition G6PD deficiency which Vasilli was born with. When he was sent to the hospital two days later, his bilirubin levels were off the charts. His mother said that when he was sent to the hospital, he was provided with phototherapy but went into cardiac arrest and had to be resuscitated.
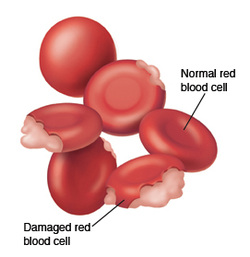
By the time he was 48 hours old, he was sleeping excessively. ‘As jaundice progresses, it has a tranquiliser-like effect on the brain because bilirubin — which is toxic to brain cells — starts to leak into it,’ says Dr Alexandrou. ‘The baby will sleep more than a newborn should, it may seem lethargic and will not cry for feeds every three to four hours as a normal newborn will do.’ Even at this stage, if caught in time, phototherapy (which involves putting the baby under a fluorescent light) can help as it makes the bilirubin more soluble and therefore easier to flush out of the body. However, if left untreated, lasting damage is caused to the brain. ‘Affected babies often lose their ability to move and they also develop uncontrollable movements of the body and hearing loss,’ says Dr Alexandrou. ‘Once the damage is done, there is no way it can be reversed.’ Pathophysiology of G6PD deficiency: Hemolytic anemia is the most common medical problem that people with glucose-6-phosphate dehydrogenase deficiency are associated with. This happens when the red blood cells are destroyed at a much faster rate than which the body is able to replace them. This is because G-6-PD is associated in supplying reducing energy to cells by maintaining the levels of the reduced form of the co-enzyme, nicotinamide adenine dinucleotide phosphate (NADPH), as it is the rate-limiting enzyme that converts glucose-6-phospate into 6-phosphoglucono-δ-lactone which supplies the reducing energy. Hence, NADPH maintains the supply of reduced glutathione in the cells which eliminates free radicals that are responsible for causing oxidative damage. As the only source of reduced glutathione in red blood cells is from the G-6-PD / NADPH pathway, the red blood cells which carries oxygen are at a risk of damage from oxidizing free radicals as the protective effect of glutathione is reduced. Therefore people with G-6-PD deficiency have a higher risk of hemolytic anemia in states of haemolytic stress. Individuals suffering from this type of anemia may experience effects such as rapid heart rates, shortness of breath, yellowing of the skin and whites of the eyes or jaundice, paleness, dark urine and fatigue. The trigger for the hemolytic anemia in people with G-6-PD deficiency are often bacterial or viral infections as well as certain drugs like medications involved in treating malaria and some antibiotics. Additionally, consumption of fava beans, which are oxidants, or inhalation of fava plant pollen can also trigger the haemolytic anemia. In severe cases, people with G-6-PD deficiency may experience acute renal failure. This is because in conditions of oxidative stress, all the remaining glutathione is consumed. As a result, proteins such as haemoglobin and enzymes are damaged by the oxidants which leads to electrolyte imbalance, cross bonding and protein deposition in the red cell membranes. Damaged red blood cells are then phagocytosed and sequestered in the spleen. Hemoglobin is then either metabolised to bilirubin which causes jaundice at high concentrations or excreted by the kidney in severe cases, causing acute renal failure. Content references:
http://ghr.nlm.nih.gov/condition/glucose-6-phosphate-dehydrogenase-deficiency http://en.wikipedia.org/wiki/Glucose-6-phosphate_dehydrogenase_deficiency http://www.doctorslounge.com/hematology/diseases/g6pd.htm Picture references: http://www.vanderbilthealth.com/includes/healthtopics/article.php?ContentTypeId=3&ContentId=40319&Category=Hematology%20and%20Blood%20Disorders&SubtopicId=33128&lang=en§ion=33113 http://www.joy2taste.com/store/289/palace-deli-salted-broad-beans-in-ribbon-stripe/ http://www.rehcfl.com/blog/patient-education/do-you-have-symptoms-of-kidney-failure-symptoms-occur-when-your-kidney-function-is-less-than-15/ Glucose-6-phosphate dehydrogenase (G6PD) is an enzyme that is present in every single cell. It is responsible for catalyzing the first reaction of the pentose phosphate pathway where glucose-6-phosphate is oxidized to 6-phosphogluconolactone.
During this reaction, nicotinamide adenine dinucleotide phosphate (NADPH) is produced and it is responsible for multiple reductive biosynthetic reactions such as the regeneration of the reduced form of glutathione (GSH). Glutathione peroxidase in erythrocytes uses GSH to detoxify hydrogen peroxide by converting it to water. This important process is heavily reliant on the production of NADPH from the pentose phosphate pathway. Without NADPH, oxidized glutathione cannot be reduced to GSH which is required in the detoxification of hydrogen peroxide. Therefore, G6PD deficiency in these cells will result in the decreased production of NADPH as this is the only NADPH-producing reaction which will lead to lower amounts of GSH in red blood cells. As red blood cells are highly sensitive to oxidative damage, they will be destroyed as they will not be able to detoxify hydrogen peroxide. Dean Holder is a 19 year old student living with his mother in West London. He has always been interested in film-making and photography. On the outside, he is just an ordinary teenager. However, he has a condition called G6PD deficiency. Let’s see how he dealt with this condition in the documentary! Video reference:
https://www.youtube.com/watch?v=DiGGwn1WE_A 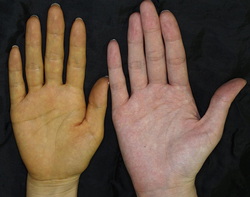 1. Symptoms: Some of the symptoms, which are more common in men, includes: - Yellow colouring of the skin - Dark yellow-orange urine - Fatigue -Pallor (unhealthy pale appearance) and general deterioration of physical conditions - Heavy and fast breathing - Weak and rapid pulse - Enlarged spleen  2. Diagnosis: General tests: Bilirubin level → measure amount of bilirubin Complete blood count, including red blood cell count → measure amount of RBC Specific tests: Phenotypic Diagnostic Test: This test measures the qualitative and quantitative amount of enzymatic activity by G6PD. Dyes or fluorescent markers are used to indicate the rate of G6PD enzyme in reducing NADP to NADPH. Spectrophotometry is used to detect the conversion of NADP to NADPH. Molecular Diagnostic Test: This test identifies the absence or presence of mutations to the G6PD gene itself. Primers specific to the G6PD gene are used. It overcomes the problems of the phenotypic diagnostic test such as varying enzymatic activity and reagent breakdown. Content references:
http://www.nlm.nih.gov/medlineplus/ency/article/000528.htm http://www.map.ox.ac.uk/client_media/publications/Howes_G6PDDef_AdvParasit.pdf http://www.nlm.nih.gov/medlineplus/ency/article/000528.htm Picture references: http://www.jaad.org/article/S0190-9622(12)00248-4/fulltext http://www.reddit.com/r/gifs/comments/22wm1q/heavy_breathing_cat_animated_oc/ http://www.webmd.boots.com/women/ss/slideshow-thyroid-symptoms-and-solutions |